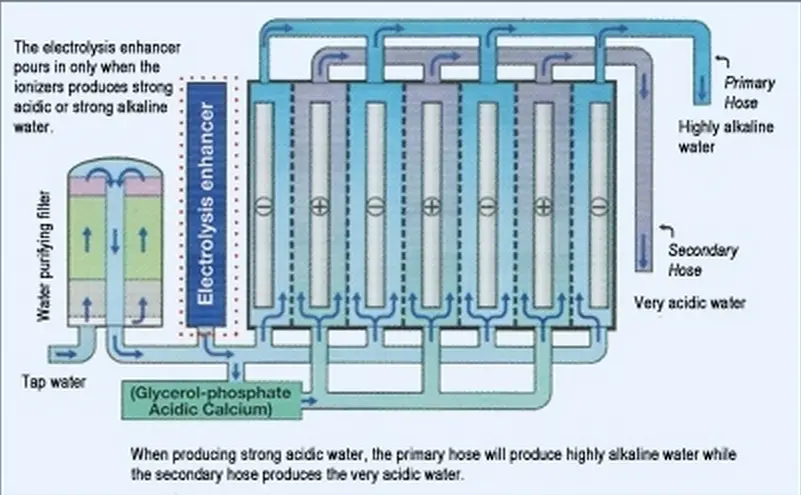
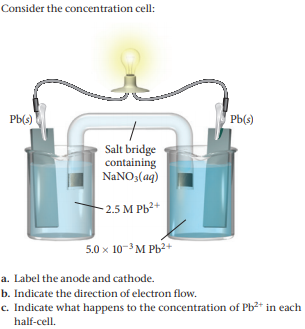
Since this style of capacitor is polarized (they have to be placed in a certain orientation) they must have indicating marks on the PCB to help determine how they should be placed. The equation we wrote above describes silver ions gaining electrons to form solid silver so we are reasonably confident that our answer is correct.When designing footprints for electrolytic capacitors it is important to place clear indicating marks to show the components’ orientation. Silver ions, Ag +, gain electrons to form metallic silver, Ag (s), at the cathode. Something must happen to the silver at the cathode. Oxidation occurs at the anode, so Mg (s) loses electrons to form Mg 2+ at the anode. The oxidation of magnesium metal to produce magnesium ions will therefore occur spontaneously. Therefore magnesium metal is a stronger reductant (reducing agent) than silver metal, that is, it is easier to oxidise magnesium metal than it is to oxidise silver metal. Magnesium is a very active (reactive) metal, much more active (reactive) than silver. Write the equation for the reduction of Ag +.Reduction (gain of electrons) occurs at the cathode.Īg + gains 1 electron to form Ag (s) at the cathode. Mg (s) loses 2 electrons to form Mg 2+ at the anode. Oxidation (loss of electrons) occurs at the anode. The two solutions are connected by a salt bridge. The two electrodes are connected by an external wire. In the other half cell, a solid zinc electrode is placed in 1 mol L -1 aqueous solution of zinc sulfate. In one half-cell a solid copper electrode is placed in 1 mol L -1 aqueous solution of copper(II) sulfate. The standard Daniell Cell is a galvanic cell (or voltaic cell) composed of two half-cells.
Cathode anode diagram free#
No ads = no money for us = no free stuff for you! Worked Example : Cell Diagram for the Daniell Cell The cell diagram can also be written using the chemical formula for each electrolyte.įor example, if the electrolyte in each half-cell in the example above was a nitrate solution, the cell diagram could be written as : Which is the cell diagram for this galvanic (voltaic) cell.

Double vertical lines, ||, indicate the connection between the two electrolyte solutions such as a salt bridge.(for example, a solid electrode in contact with a solution) Single vertical lines, |, indicate a phase boundary or junction.The usual convention for writing a cell diagram is: You need to become an AUS-e-TUTE Member! Cell Diagrams for Galvanic Cells (Voltaic Cells) Chemistry Tutorial Key ConceptsĪ cell diagram is Chemistry's short-hand for representing a galvanic cell (voltaic cell). Want chemistry games, drills, tests and more? Galvanic Cell Diagrams Chemistry Tutorial More Free Tutorials Become a Member Members Log‐in Contact Us
